KPV peptide guide: what the preclinical research shows
KPV is the three-amino-acid fragment of alpha-MSH. Preclinical data points to NF-kB suppression, gut and skin effects, but human trials remain absent.
KPV peptide is the three-amino-acid C-terminal tail of alpha-melanocyte-stimulating hormone, isolated from a 13-residue parent that has been studied as an anti-inflammatory regulator since the 1990s. Preclinical evidence has tracked KPV across mouse colitis models, human skin permeation studies, and bronchial epithelial cell cultures. As of 2026, no registered human clinical trials for KPV exist. The honest version of this guide names that gap upfront, then walks through what the animal and cell-culture data actually show.
KPV is the C-terminal three amino acids of alpha-MSH, not a standalone novel peptide#

The active sequence of KPV peptide is lysine-proline-valine: three residues in a row, written as Lys-Pro-Val or one-letter K-P-V. These three amino acids are not arbitrary. They correspond to positions 11, 12, and 13 of alpha-melanocyte-stimulating hormone, a 13-residue neuropeptide that the body produces as a post-translational cleavage product of pro-opiomelanocortin in the pituitary, skin, and immune cells. The full alpha-MSH molecule has been studied as an anti-inflammatory and immunomodulating signal across more than three decades of literature (Catania et al., PMC2095288).
The key observation that drives the KPV literature is this: when researchers cleaved the parent alpha-MSH down to its C-terminal three residues, the anti-inflammatory potency was retained, in many assays at comparable or higher levels than the full peptide. The melanocortin receptor binding, on the other hand, was lost. Alpha-MSH binds the MC1R, MC3R, MC4R, and MC5R family because of pharmacophore residues elsewhere in the chain. KPV does not have those residues. The practical consequence is that lysine-proline-valine peptide retains the anti-inflammatory signalling without producing the skin-darkening, sexual-function, or appetite effects associated with full melanocortin receptor agonism.
This structural framing of KPV as an alpha-MSH fragment peptide matters because it separates KPV from the rest of the peptide landscape Klarovel covers. BPC-157 is a 15-amino-acid synthetic fragment of a gastric protein. TB-500 is a 17-amino-acid fragment of thymosin beta-4. KPV is three amino acids. The size has practical implications: shorter peptides face fewer enzymatic cleavage sites, can survive oral and topical routes that destroy larger molecules, and reach intracellular targets at concentrations the larger peptides cannot. Whether these implications translate to human therapeutic benefit is what the rest of this guide examines.
KPV blocks NF-kB without engaging melanocortin receptors#
The mechanism of KPV is intracellular, not receptor-mediated in the classical sense. Research has shown KPV is transported into intestinal epithelial cells and immune cells by PepT1, a proton-coupled di- and tripeptide transporter whose expression rises in inflamed gut tissue (Dalmasso et al., PubMed 18061177). PepT1 normally handles dietary di- and tripeptides during digestion. KPV's three-residue size makes it a substrate. Once inside the cell, KPV interferes with NF-kB activation, the master switch for pro-inflammatory gene transcription.

The downstream cytokine effects fall in line with NF-kB suppression. Studies have shown KPV reduces TNF-alpha, IL-6, IL-1-beta, IL-12, and IFN-gamma at the mRNA and protein level in stimulated cell lines and murine inflammation models (Kannengiesser et al., PubMed 18092346). Nanomolar concentrations produce the effect; the active range is two to three orders of magnitude below where most peptides need to sit to register on assays. In a Cooper et al. paper using human bronchial epithelial cells, KPV's anti-inflammatory action was observed with an MC3R-related context, suggesting that some target tissues retain receptor-level interaction even when the gut-epithelial picture is purely intracellular (Cooper et al., PMC3403564).
The receptor-independent piece of this mechanism is one of the reasons KPV appears repeatedly in alpha-MSH fragment peptide literature as a target for inflammation research. A full alpha-MSH dose produces measurable melanocortin receptor activation, with the cosmetic and physiological effects that follow. KPV does not. For research applications targeting inflammation specifically, the fragment is cleaner: anti-inflammatory action with the receptor activity stripped out.
The strongest KPV evidence sits in animal colitis models, not humans#
This is the post's honesty fulcrum. As of 2026, no registered human clinical trials for KPV exist on ClinicalTrials.gov. The evidence base that vendor pages and clinic-marketing blogs reference traces back, almost entirely, to mouse colitis models and isolated cell-culture work. None of it has been replicated in a controlled human trial.
The flagship preclinical paper is Dalmasso et al., 2008, published in Gastroenterology. The group used DSS-induced and TNBS-induced colitis in mice, two standard models for inducing inflammatory bowel disease-like pathology. Oral KPV at low doses reduced weight loss, decreased myeloperoxidase activity (a neutrophil infiltration marker), normalised histological inflammation scores, and lowered pro-inflammatory cytokine mRNA. The mechanism trace ran through PepT1 expression: KPV was transported into colonocytes and immune cells via the same di-/tripeptide transporter that handles dietary fragments.
A complementary line of work focused on delivery. The native KPV tripeptide degrades quickly in the small intestine before it can reach the colonic mucosa. Xiao and colleagues, 2017, published in Molecular Therapy, addressed this with hyaluronic-acid-functionalised polymer nanoparticles roughly 270 nm in diameter that carried KPV through the upper gut and released it at colonic CD44-expressing macrophages and inflamed epithelium. In their DSS-colitis model, the targeted nanoparticles accelerated mucosal healing and reduced colon-shortening, weight loss, and cytokine elevation more than free KPV. The Kannengiesser review of melanocortin-derived tripeptides positioned this work as the most credible delivery-side advance for KPV in inflammatory bowel disease research (Kannengiesser, PubMed 18092346).
What the literature does not show is a phase 1 safety trial in humans, a phase 2 dose-finding trial, or a phase 3 efficacy trial. The clinical experience that vendor sites cite is practitioner observation in research settings, not regulated clinical evidence. KPV's relationship to ulcerative colitis and Crohn's disease is preclinical association, not validated human therapy. The honest framing for readers researching KPV peptide ulcerative colitis evidence: preliminary evidence indicates anti-inflammatory effects in mouse models that resemble the diseases, with no human trial yet completed.
For readers also evaluating cousin tissue-healing peptides, the BPC-157 complete guide walks through a longer animal-evidence base and a smaller but non-zero set of human case reports, and the TB-500 complete guide covers a thymosin-beta-4 fragment with similarly preclinical-weighted evidence.
KPV's other research applications target skin and wound healing#
The second arm of KPV research is topical and transdermal, a natural extension of the parent alpha-MSH's role in skin biology. Multiple preclinical lines of work have studied KPV in dermatological inflammation, wound healing, and cosmetic delivery. The literature here is smaller than the gut-inflammation track, but informative.
The most carefully measured of these papers is the Pawar and Banga group's work on transdermal iontophoretic delivery of KPV across microporated human skin, published in the Journal of Pharmaceutical Sciences in 2017 (ScienceDirect). The team measured KPV permeation across excised human skin under four conditions: passive diffusion alone (below detection threshold), microneedle treatment alone (a measurable 4.4 micrograms per square centimetre per hour), iontophoresis added to microneedles (an 8-fold increase over microneedles alone), and combined iontophoresis plus microneedles (a 35-fold increase over microneedles alone). The pH dependence mattered: KPV carries a positive charge below physiological pH, which is why anodal iontophoresis works and which constrains the formulation pathway any future human topical product would have to follow.
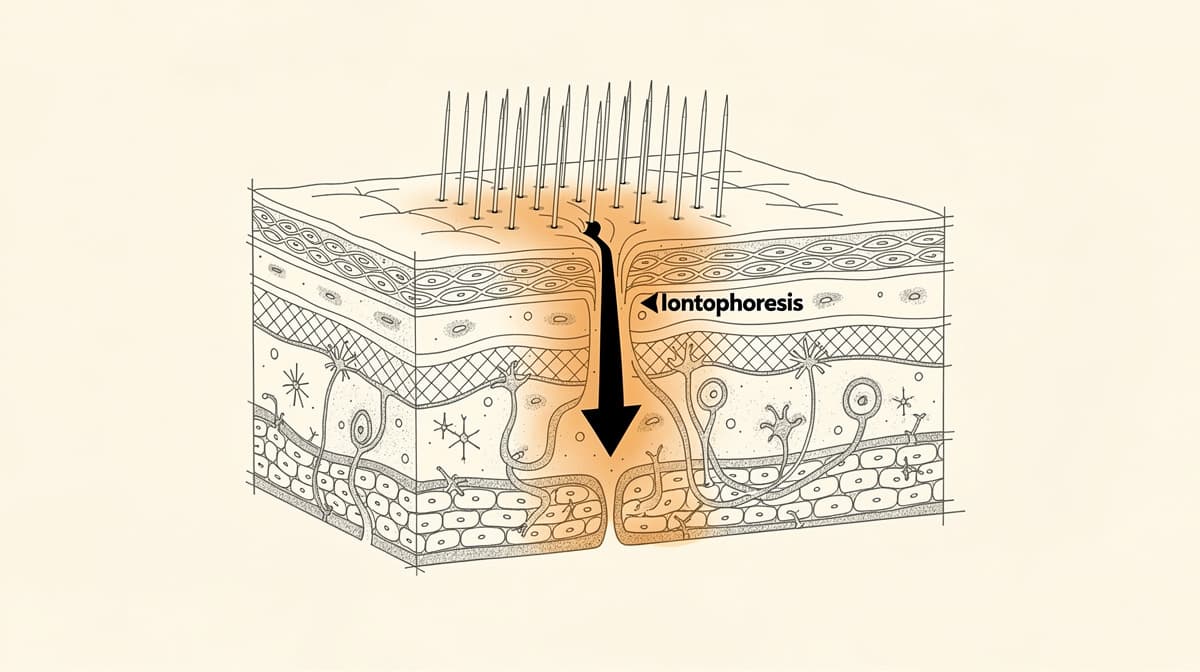
Wound-healing data tracks animal models more than human trials. Older work has reported faster epithelial closure and reduced inflammation around full-thickness skin wounds in rabbits and rodents treated with KPV-containing formulations. The patent literature on dermatological disorders has captured comparison studies for skin-condition applications; these are exploratory, not validated. Research has shown KPV's anti-inflammatory action in cell-culture models of contact dermatitis and atopic-dermatitis-like inflammation; the receptor-independent intracellular pathway documented in gut tissue appears to operate similarly in skin.
The honest framing for the topical arm: KPV is associated with anti-inflammatory and pro-healing effects in animal and ex-vivo human-skin models, but no validated human dermatological indication exists. Readers comparing peptide-based topical research should also see the GHK-Cu complete guide, which covers a copper-binding tripeptide with the deepest human cosmetic evidence base of any peptide in the Klarovel catalog.
Research dosing for KPV runs 200 to 500 mcg per administration, with no clinical anchor#
The dose discussion is the section where compliance language has to do real work. Research protocols for KPV, captured in the Klarovel peptide_research catalog and consistent across the practitioner literature, describe a range of 200 to 500 mcg per administration via subcutaneous injection or oral capsule, taken once daily, in cycles of 8 weeks on followed by 4 weeks off. None of these doses have been validated in a registered human clinical trial. They are practitioner-derived ranges, not clinical-trial-derived ranges.
The KPV peptide dosage discussion needs its caveats stated cleanly. Practitioner-derived ranges work as a starting reference for research-context use, but they are not equivalent to the dose-finding evidence behind an FDA- or EMA-approved drug. A typical approved drug carries a known maximum tolerated dose, a known dose-response curve, and a known therapeutic window with margins on either side. KPV does not. The 200-to-500-mcg range reflects what practitioners reported using, not what controlled trials have shown to be optimal or safe at the upper end.
The KPV peptide side effects that have been reported in research-context use are mild and self-limiting: occasional injection-site reactions for the subcutaneous route, mild gastrointestinal complaints for oral use, and rare headache. None of these have been characterised in a controlled trial, which means rare events that affect 1 in 10,000 patients would not yet be visible. For an anti-inflammatory peptide with the receptor-cleaner profile of KPV, the absence of accumulated severe adverse-event reports is a positive signal, but the absence is also small. The research record is shallow because the research itself is shallow. Readers building broader context should review peptide contraindications for the structural categories Klarovel applies across all research-context use.
KPV is unapproved in every major jurisdiction, including Norway#
The regulatory picture for KPV is thin. The FDA has not approved KPV for any indication. In April 2026, the FDA announced changes to the status of 12 peptides under bulk-substance review; KPV's nominator withdrew its Category 2 nomination, with a Pharmacy Compounding Advisory Committee review of the 503A bulks list scheduled for July 2026. The European Medicines Agency has issued no monograph for KPV. Norway's Direktoratet for medisinske produkter (DMP) has no marketing-authorisation record for the compound. No approved therapeutic indication exists anywhere in the world as of April 2026.
The practical Norwegian framing is the same one Klarovel applies to other unapproved peptides. KPV without a Norwegian marketing authorisation falls under the same forskrift architecture that prohibits postal import of unauthorised medicines, with Tolletaten enforcing at customs. The compound's research-only positioning is documented, and readers researching it for any application that touches their own health should treat the absence of human trials as the dominant fact.
For more on the regulatory framing Klarovel applies across its entire research-positioning posture, see Klarovel's disclosures. The structural point matters: a peptide with promising preclinical data, no registered human trials, and no approved indication is not the same thing as a peptide with established human safety and efficacy, and Klarovel's editorial framing keeps that distinction visible.
Frequently asked questions about KPV peptide#
What is KPV peptide used for? KPV, the lysine proline valine peptide derived from alpha-MSH, is researched primarily for its anti-inflammatory effects in animal and cell-culture models. The strongest preclinical evidence sits in mouse colitis models, where oral KPV reduced inflammation markers in DSS- and TNBS-induced disease. A second arm of research targets skin inflammation, wound healing, and transdermal delivery. As of 2026, no human clinical trial has been registered for KPV, and no regulatory body has approved it for any indication. KPV peptide benefits documented in the literature are preclinical, not clinical.
Is KPV peptide safe? The published preclinical and research-context experience reports mild and self-limiting side effects: occasional injection-site reactions, mild gastrointestinal complaints with oral use, and rare headache. No registered human clinical trial has characterised KPV's safety profile in a controlled setting. Rare adverse events that affect 1 in 10,000 patients would not yet be visible in the research-context record. The regulatory absence (no FDA, EMA, or DMP authorisation) means there is no validated safety dossier the way there is for an approved drug.
How is KPV peptide taken? Research protocols describe two main routes: subcutaneous injection and oral capsule. The oral route is unusual for peptides, but KPV's three-residue size lets it survive enough of the upper-gut environment to reach the colon, particularly when delivered in protective formulations. Topical and transdermal formulations exist in research but are not standardised; the iontophoresis-microneedle work in human skin in vitro showed a 35-fold flux increase over microneedles alone, which is encouraging mechanistically but distant from a validated topical product.
Does KPV help with ulcerative colitis? Preclinical evidence indicates KPV reduces inflammation in mouse models of ulcerative colitis (the DSS model) and Crohn's-like inflammation (the TNBS model). These mouse-model results are not the same as human therapeutic evidence. As of 2026, no registered human clinical trial has tested KPV in ulcerative colitis, Crohn's disease, or any other inflammatory bowel disease. Patients with active inflammatory bowel disease should treat the existing literature as preclinical-only and rely on their gastroenterologist for evidence-graded therapy decisions.
Can KPV be stacked with BPC-157? Practitioner protocols sometimes pair the two, on the rationale that BPC-157 contributes broader connective-tissue and gastrointestinal mucosal effects while KPV adds anti-inflammatory action through the NF-kB pathway. The mechanisms are non-overlapping. No published human trial has tested the combination. Klarovel's protocol engine evaluates stacking compatibility automatically based on questionnaire data and known mechanism overlap; the standard caution is that stacking peptides multiplies the side-effect surface even when each is individually well tolerated. The BPC-157 complete guide covers the cousin peptide's evidence base in detail.
How long does it take KPV to work? The animal-model data shows measurable inflammation-marker reduction within days of oral or subcutaneous KPV administration in DSS colitis and similar models. Whether the same time-course holds in humans is not characterised. Research-context users typically describe gradual rather than dramatic effects, with the most-reported framing being a slow shift over weeks rather than an acute response over hours. The honest framing for prospective readers: research suggests subtle, gradual effects in animal data, with no human time-course evidence available.
Where KPV peptide fits relative to Klarovel's broader tissue-healing framework#
The honest framing for KPV peptide is the one this guide opened with. The mechanism is real, intracellular, receptor-independent in the gut and partly receptor-mediated in airway tissue, with consistent NF-kB suppression at nanomolar concentrations. The animal-model evidence in colitis and skin inflammation is real. The human-trial record is empty. The regulatory record is empty. None of these facts cancel each other out, and any guide that rounds the picture in either direction is simplifying.
Readers placing KPV peptide alongside the rest of the tissue-healing cluster should treat the comparison as one of evidence depth rather than mechanism uniqueness. BPC-157 has more animal evidence and a small human case-report base. TB-500 has the longest tissue-repair history. GHK-Cu has the deepest human cosmetic literature. KPV has the cleanest receptor-independent anti-inflammatory profile and the smallest human evidence base. For readers wanting to understand which research-context peptide class might align with their specific bloodwork, goals, and constraints, the Klarovel questionnaire is the route through. Complete the Klarovel questionnaire to see what the engine recommends from a full health profile rather than a single compound's reputation.
Keep reading

Peptides for Muscle Growth: What the Data Says
Trials report 1.1 to 1.3 kg lean mass gain on growth-hormone peptides over 6 to 12 months. The data and what it does not show, plus WADA status.

Peptides for Weight Loss: What the 2026 Trial Data Says
Four peptide medications are FDA-approved for weight loss in 2026. Trial data, mechanism, side effects, and how the approved options compare.
Selank Peptide: Russian-Approved, Not FDA-Authorized
Selank peptide is a Russian-approved anxiolytic that modulates GABA-A and enkephalins. The mechanism, the 62-patient trial, and 2026 regulatory reality.