Melanotan 2 in Norway: Law, Risks, and Safer Tan Options
What research shows about Melanotan 2, why DMP has not approved it in Norway, the documented melanoma case reports, and evidence-based alternatives for pigmentation goals.
A bronzed Instagram clip, a discreet nasal spray, and a parcel from an overseas vendor. That is the dominant pathway by which Melanotan 2 reaches Norwegian skin in 2026, and it is also the pathway that the Direktoratet for medisinske produkter (DMP) has formally moved to shut down. This guide explains what the peptide actually is, what the published research has shown, where the legal line sits, and what the credible alternatives look like.
Key takeaways#
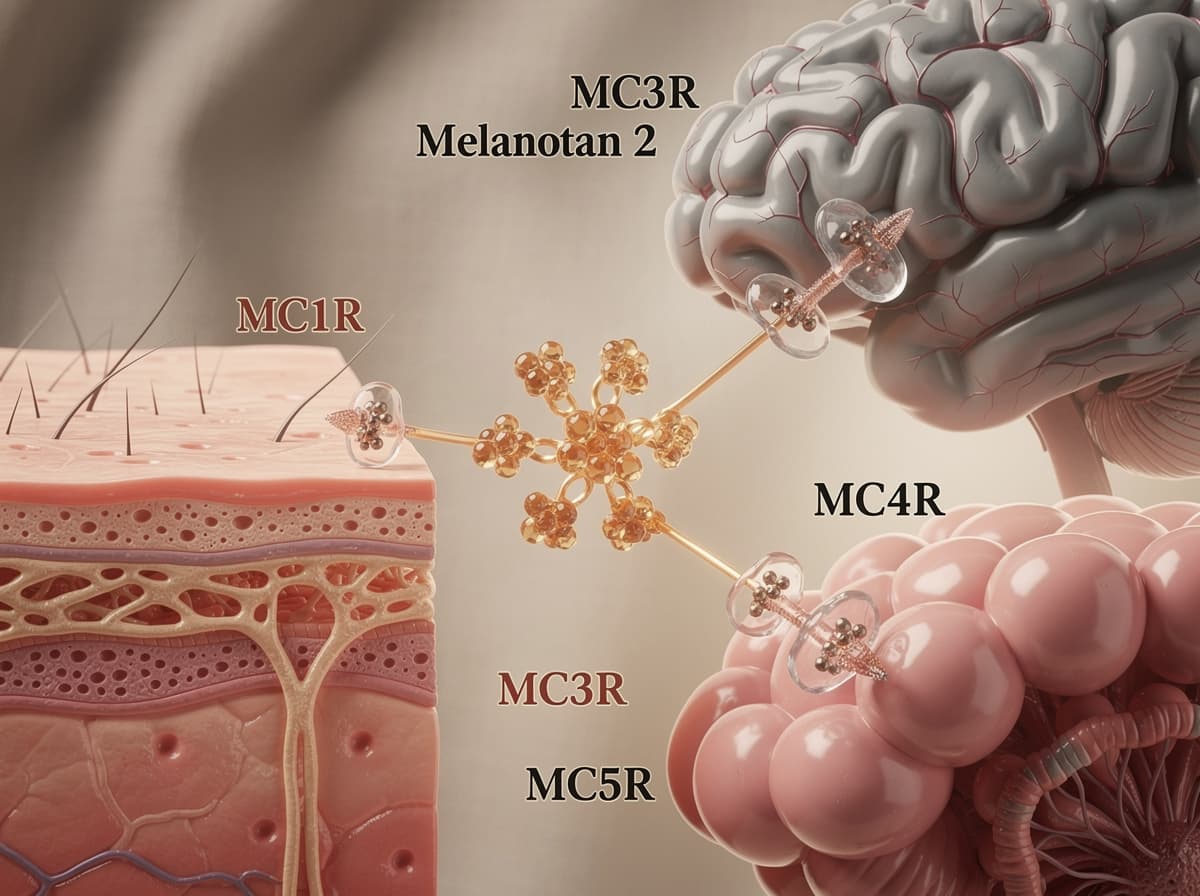
- Melanotan 2 (MT-II) is a synthetic, non-selective melanocortin receptor agonist developed at the University of Arizona in the 1980s and 1990s; it activates MC1R, MC3R, MC4R, and MC5R.
- It is not approved as a medicine in Norway or any other country, and DMP issued a 2026 enforcement decision against domestic advertising of melanotan and retatrutide.
- Documented side effects include nausea, facial flushing, spontaneous erections, darkening of moles, and at least four published case reports of melanoma diagnosed during or shortly after use.
- A regulated relative, afamelanotide (Scenesse), is EMA-approved since December 2014 for erythropoietic protoporphyria, but only via specialist centres, not for cosmetic tanning.
- Research-grade Melanotan 2 is supplied by specialised vendors strictly for laboratory use; Klarovel does not stock or sell peptides.
Melanotan 2 is a non-selective melanocortin agonist, not a tanning supplement#
The molecule itself is a cyclic heptapeptide. Melanotan II acts as a non-selective agonist of the melanocortin receptors MC1, MC3, MC4, and MC5. The skin-darkening effect that drives consumer demand comes from MC1R activation on melanocytes. Melanotan II produces melanogenesis by activation of the MC1 receptor, whereas its clinically documented sexual effects are thought to be related to its ability to activate the MC4 receptor. Other effects, mostly regarded as adverse effects, include flushing, nausea, vomiting, stretching, yawning, and loss of appetite.
The compound was never a wellness supplement. Research in the early 1960s showed that in rats, administration of α-MSH caused sexual arousal, and work on this continued in many labs up through the 1980s, when scientists at the University of Arizona began attempting to develop α-MSH and analogs as potential sunless tanning agents, and synthesized and tested several analogs, including melanotan-I and melanotan II. The original pharmaceutical track for MT-II was erectile dysfunction research, which preliminary evidence supported in a small placebo-controlled trial where the peptide led to penile erection in 17 of 20 men but also produced severe nausea at higher doses.
Norwegian regulators have classified Melanotan as an unlicensed medicine#
The legal framing in Norway is unambiguous. The substance is not approved as a medicine in Norway and is illegal to import. A senior physician at DMP, Sara Viksmoen Watle, has warned against use because it may be dangerous and may increase the risk of skin cancer.
In January 2026, DMP escalated from public warnings to formal enforcement. According to the DMP decision document, advertising is only permitted for products holding a Norwegian marketing authorisation, and advertising for retatrutide and melanotan is therefore not permitted, as neither holds marketing authorisation in Norway. The same decision confirms that Melanotan is a medicine that stimulates skin cells (melanocytes) to produce more pigment and is used to achieve darker skin colour, and is not an approved medicine in Norway.
Customs enforcement is active. The figures show that the driver stopped at Svinesund is far from the only one trying to bring the substance into Norway. Most shipments of melanotan are not seized but destroyed. The difference is whether the importer is reported for the import or not.
The melanoma signal is small but real#
The mechanistic concern is straightforward: a drug that drives melanocyte proliferation in skin that may already carry atypical nevi is the kind of exposure dermatologists treat seriously. Research has shown that the published case-report literature is thin but consistent. The 2014 paper by Hjuler and Lorentzen in Dermatology is the most cited example. A 2025 ScienceDirect case report adds a mucosal melanoma signal tied to nasal-spray administration: risk factor analysis suggested the most likely cause of the mucosal melanoma in this healthy young patient was related to the use of Melanotan II nasal spray, particularly as oral pigmentation began soon after its initiation, highlighting a unique instance of mucosal malignant melanoma potentially associated with Melanotan II.
Dermatology experts have repeatedly described the same pattern. Case reports have described melanomas emerging from existing moles either during or shortly after the use of Melanotan-II, however evidence for causal associations is lacking and definitive proof is yet to be established. Preliminary evidence also points to other longer-term changes: there is concern that melanotan II may increase the risk of melanoma, deepening of the colour of moles, new moles and atypical melanocytic naevi, melanonychia (brown to black discolouration of one or more nails), and rhabdomyolysis (potentially fatal destruction of muscle cells).
The honest summary is that causation has not been proven in a controlled study, but the signal is plausible enough that every dermatology body that has reviewed the literature recommends against cosmetic use.
Side effects are common, immediate, and not trivial#
The acute side-effect profile is well characterised even outside the melanoma question. A University of Arizona human study of MT-II in men with erectile dysfunction documented that nausea and yawning were frequently reported side effects due to Melanotan II; at a dose of 0.025 mg/kg, 12.9% of subjects had severe nausea. DermNet's clinical summary lists additional acute reactions associated with the peptide: during clinical trials for its use as a tanning agent, melanotan II was found to be a potent stimulator of male erections, and a new drug based on melanotan II, bremelanotide, was developed to take advantage of this property.
For men, priapism is the most acute risk. Studies have shown that prolonged erections lasting hours after a single dose are a recurrent reason for emergency-department presentation, with BMJ Case Reports documenting Melanotan-induced priapism episodes. The product-quality layer compounds everything else. Buyers do not know what they are putting into their bodies when ordering goods illegally online, and bottles seized at Svinesund had no ingredient list.

Afamelanotide is the regulated cousin, and it is not available for cosmetic use#
The distinction between Melanotan 2 and afamelanotide (Melanotan 1, brand name Scenesse) matters because it is often deliberately blurred by online sellers. Afamelanotide is research-validated and regulated. The EMA authorisation record confirms that Scenesse contains the active substance afamelanotide, is only prescribed by specialist doctors in recognised centres for treating EPP, and should only be used by doctors who have been properly trained, with one implant injected under the patient's skin once every 2 months.
The pivotal evidence comes from the New England Journal of Medicine trial, which has been shown to be the basis for both EMA and FDA approval. Two multicenter, randomized, double-blind, placebo-controlled trials of subcutaneous implants containing 16 mg of afamelanotide were conducted, with 74 patients in the European Union and 94 patients in the United States randomly assigned 1:1 to receive an implant every 60 days. Studies have shown afamelanotide is associated with measurable photoprotection in a defined patient group, but the regulatory framework explicitly excludes cosmetic indications. The FDA review for Scenesse spelled this out: the agency required risk management to address theoretical risks of melanoma, hypersensitivity reactions, implantation administration errors, and off-label use for aesthetic purposes.
Translation: even the regulated, MC1R-selective version of this chemistry was approved with melanoma as a flagged theoretical risk and aesthetic off-label use as something regulators actively did not want to encourage.
Quality and contamination are the unspoken risks#
Even if a Norwegian buyer accepts the legal and pharmacological risks, the supply chain itself is a second-order hazard. A 2015 vendor analysis cited across the dermatology literature found that some products contained only 43% of the labeled melanotan-2. Practical purity verification is non-trivial for a private buyer: HPLC certificates are easy to fake, and nasal-spray formulations sold to consumers bypass even the minimal sterility controls of a lyophilised vial.
This is why research-grade peptide chemistry exists on a different track entirely. Specialised laboratory suppliers handle MT-II strictly for Research Use Only contexts with chain-of-custody documentation. Klarovel does not stock, sell, or fulfil peptides; the protocol layer we curate is built around evidence review, dose modelling in the peptide calculator, and editorial guidance, not product distribution. Anyone evaluating peptide research should read our how it works page and the disclosures before going further.
Safer routes to the pigmentation outcome users actually want#
The honest reframe is that most Melanotan 2 users do not want melanocortin agonism; they want skin a few shades darker without UV damage. Three categories already deliver that without the regulatory and oncologic exposure:
-
DHA-based self-tanners. Self-tanners typically contain dihydroxyacetone (DHA), an ingredient that reacts with amino acids in the outermost layer of the skin to create a temporary darkening effect, and the process does not involve UV radiation and does not damage living skin cells.
-
Tyrosinase-pathway support topicals. Niacinamide and certain peptide cosmetics modulate melanin distribution without systemic melanocortin activation. The evidence base is more modest than DHA but the safety profile is excellent.
-
Pigment-targeted dermatology procedures for users whose underlying goal is even tone rather than overall darkening. These belong in a clinic conversation, not a forum.
For users specifically interested in the pigmentation-research cluster from the molecular angle rather than the cosmetic one, the Klarovel register flow opens access to the protocol library where evidence-graded options sit alongside the safety annotations.
Frequently asked questions about Melanotan 2 in Norway#
Is Melanotan 2 illegal to buy in Norway?
Yes. The substance is classified as a medicine under the Norwegian Medicines Act and has no marketing authorisation, which means private import is prohibited and customs routinely destroys or seizes shipments. Melanotan is regarded as a medicine under the Medicines Act § 2. The product is not sold legally in Norway, and users typically buy it online from foreign web shops. However, it is not permitted for private individuals to import medicines under the Medicines Act § 13. The substance will therefore be stopped at customs if discovered.
What is the difference between Melanotan 1 and Melanotan 2?
Melanotan 1 (afamelanotide) is a longer linear peptide that selectively targets MC1R and is approved as Scenesse for erythropoietic protoporphyria. Melanotan 2 is a shorter cyclic non-selective agonist that hits MC1R, MC3R, MC4R, and MC5R, which is why its side-effect profile is broader and why it is not approved anywhere. Melanotan 2 is a shorter cyclic variant of Melanotan 1 that was developed in the 1990s. Melanotan 2 was found to increase skin pigmentation at lower cumulative doses than Melanotan 1, however in clinical trials it was found to have more side effects including nausea, somnolence and penile erections.
Does Melanotan 2 cause melanoma?
The honest answer is that causation has not been proven, but case reports have been published and the biological mechanism is plausible. Stimulating pigment cells repeatedly can cause abnormal proliferation, which can jumpstart the progression to the possible development of melanoma, and previous case reports have shown some Melanotan-II users develop skin-based complications, including melanoma, however the evidence is still limited. Most dermatology bodies recommend baseline mole mapping and annual skin checks for anyone who has used it.
Is the nasal-spray version safer than injection?
There is no evidence supporting that. The 2025 ScienceDirect case report described an oral mucosal melanoma in a 22-year-old user of the nasal spray, with pigmentation appearing soon after initiation. The nasal route bypasses the visual feedback of injection-site darkening but introduces direct mucosal exposure, which is itself a documented melanoma-risk site.
What about athletes, is Melanotan 2 banned by WADA?
No, but the absence from the WADA list is a technical classification issue, not a safety endorsement. It is often called "barbie-drug" but does not appear on the WADA doping list because the substance does not meet enough of WADA's criteria. Norwegian sport federations and the underlying Medicines Act prohibition still apply.
Can Klarovel help me obtain Melanotan 2?
No. Klarovel does not sell, stock, or fulfil peptides, and we will not refer users to vendors of unapproved medicines. Research-grade MT-II is available from specialised laboratory suppliers for Research Use Only purposes outside Norway, but procurement, customs clearance, and any downstream consequences sit entirely with the buyer. The Klarovel role is editorial and protocol-curation, not distribution; the related-cluster reading on PT-141, the melanocortin derivative that did complete regulatory development, is a more productive next step for most readers.
The conviction#
Melanotan 2 sits in a category Klarovel exists specifically to clarify: a peptide with real pharmacology, a plausible cosmetic upside, and a regulatory and oncologic profile that has not earned the trust of any medicines authority worldwide. The Norwegian framing makes the cost-benefit even sharper, because DMP enforcement is now active and customs interdiction is routine. Readers who want the underlying pigmentation outcome have better options. Readers who want to understand the melanocortin system as a research domain should start with the peptide calculator and the editorial protocol library; create a free account at Klarovel to access the full library.
Keep reading

Are peptides legal in Norway? The 2026 regulatory answer
Peptides in Norway are mostly unlawful to import privately. What DMP stops at customs, what's approved, and how Norwegian law handles it.

Who shouldn't take peptides: evidence-based contraindications
Peptides aren't for everyone. Active cancer, pregnancy, uncontrolled diabetes, severe kidney or liver impairment: what the evidence says about who should avoid which classes.
The GH peptide axis: how GHRH, ghrelin, and IGF-1 connect
A pillar guide to the GH peptide axis: how GHRH analogs and ghrelin mimetics drive pulsatile growth hormone, and what somatopause changes.