The GH peptide axis: how GHRH, ghrelin, and IGF-1 connect
A pillar guide to the GH peptide axis: how GHRH analogs and ghrelin mimetics drive pulsatile growth hormone, and what somatopause changes.
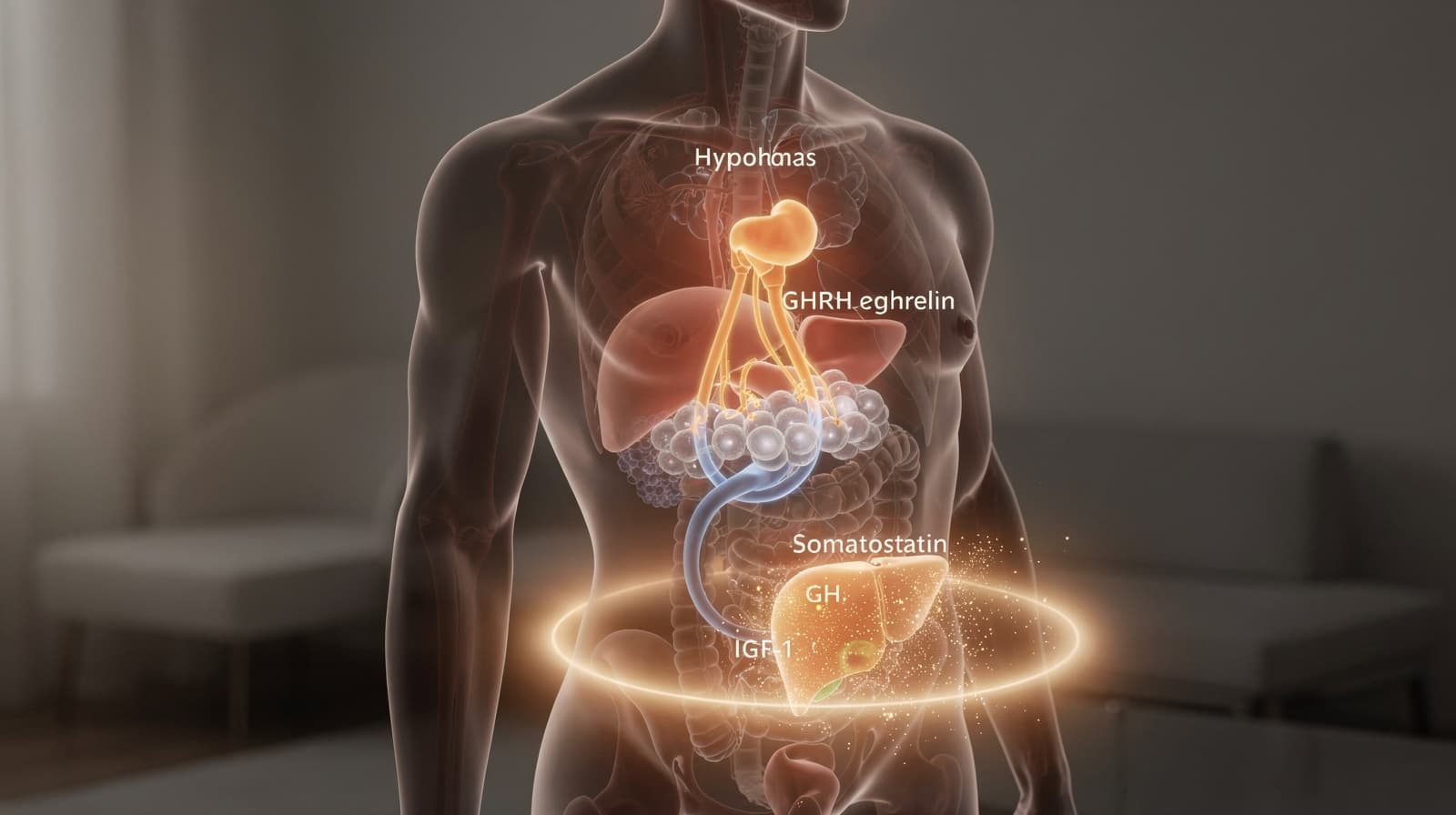
Most peptide content treats individual molecules as standalone tools. That framing hides the more useful truth: ipamorelin, CJC-1295, MK-677, and tesamorelin all act on the same regulatory circuit, and understanding that circuit predicts which compound fits which goal. This pillar walks through the GH peptide axis, the receptors involved, and how age changes the picture.
Key takeaways#
- The GH axis is governed by three inputs: hypothalamic GHRH (stimulatory), somatostatin (inhibitory), and stomach-derived ghrelin (stimulatory through a separate receptor).
- Pituitary somatotrophs release growth hormone in pulses; downstream hepatic IGF-1 closes the feedback loop by suppressing further GH output.
- Research suggests two receptor families are relevant for peptide protocols: the GHRH receptor (targeted by CJC-1295 and tesamorelin) and the GHSR-1a ghrelin receptor (targeted by ipamorelin and MK-677).
- Somatopause, the age-related decline in GH and IGF-1, has been shown to track with shifts in body composition and is associated with reduced lean mass.
- Pairing a GHRH analog with a ghrelin mimetic produces a supra-additive pulse, which is the mechanistic rationale behind common combination protocols.
The axis is a three-input control loop, not a single switch#
The classical description of growth hormone regulation, refined since the 1960s, has GHRH and somatostatin as opposing hypothalamic inputs. Pituitary synthesis and secretion of GH is stimulated by episodic hypothalamic secretion of GH releasing factor and inhibited by somatostatin, and Insulin-like Growth Factor I (IGF-I) inhibits GH secretion by a negative loop at both hypothalamic and pituitary levels.
That two-input model held until 1999, when the discovery of ghrelin added a third regulator from outside the brain. The gastric peptide ghrelin is also a potent GH secretagogue, which acts to boost hypothalamic GHRH secretion and synergize with its pituitary GH-stimulating effects. In other words, ghrelin hits a distinct receptor (GHSR-1a) and amplifies the GHRH signal both centrally and at the somatotroph.
The practical implication: any peptide that influences GH output has to act through one of these three control points. GHRH analogs push the accelerator. Ghrelin mimetics push a different accelerator. Nothing currently available in research-peptide form removes the somatostatin brake directly, which is why protocols time injections around endogenous somatostatin lows (typically before sleep or in the early morning fasted state).
GH release is pulsatile, and the pulse pattern matters more than the average#
Continuous GH exposure does not reproduce the physiology of healthy GH secretion. Studies have shown that GH secretion in man is pulsatile and this pattern is regulated by both GHRH and somatostatin, with bursts of GH secretion mediated by a reduction of tonic hypothalamic somatostatin secretion.
This is why long-acting recombinant GH and a well-designed peptide protocol produce different downstream signatures. A short GHRH pulse layered on top of an endogenous somatostatin trough preserves the pulse architecture; continuous receptor occupation does not. Research has shown the GH response to sustained GHRH infusion attenuates within hours, confirming that the somatostatin brake, not GHRH supply, dictates pulse timing.
For protocol design, three points follow:
- Timing relative to meals matters because nutrient intake suppresses GH and elevates somatostatin.
- Timing relative to sleep matters because the largest endogenous GH pulse occurs in early slow-wave sleep.
- Combining a GHRH analog with a ghrelin mimetic is associated with a larger pulse than either compound alone, because the two receptors converge on the same somatotroph through complementary action.
Two receptor families, two peptide categories#
Every peptide marketed as a "growth hormone secretagogue" falls into one of two mechanistic buckets, defined by the receptor it binds.
GHRH receptor agonists mimic endogenous GHRH at the pituitary GHRH receptor. Growth Hormone-Releasing Hormone (GHRH), secreted by neurons in the arcuate nucleus of this brain structure, stimulates HGH production in the anterior pituitary gland. CJC-1295 and tesamorelin are the two compounds most often discussed in this category. Tesamorelin is the only FDA-approved GHRH analog, studied primarily in HIV-associated lipodystrophy. Klarovel covers tesamorelin's mechanism and trial data in the tesamorelin guide.
Ghrelin receptor agonists (also called GHSR-1a agonists, growth hormone secretagogues, or "GHS") bind the receptor that endogenous ghrelin activates. Research has shown that ghrelin exerts a synergistic effect with GHRH in the induction of GH secretion ; this is the receptor that ipamorelin and MK-677 target. Ipamorelin is an injected, short-acting peptide; MK-677 (ibutamoren) is an orally bioavailable non-peptide ghrelin mimetic. The mechanistic comparison is laid out in the MK-677 guide and the ipamorelin overview.
The reason combination protocols exist is precisely because these are two different receptors with complementary downstream signaling. The paired CJC-1295 plus ipamorelin approach is covered in detail in Klarovel's CJC-1295 and ipamorelin protocol article.
Somatopause shifts the baseline that protocols try to address#
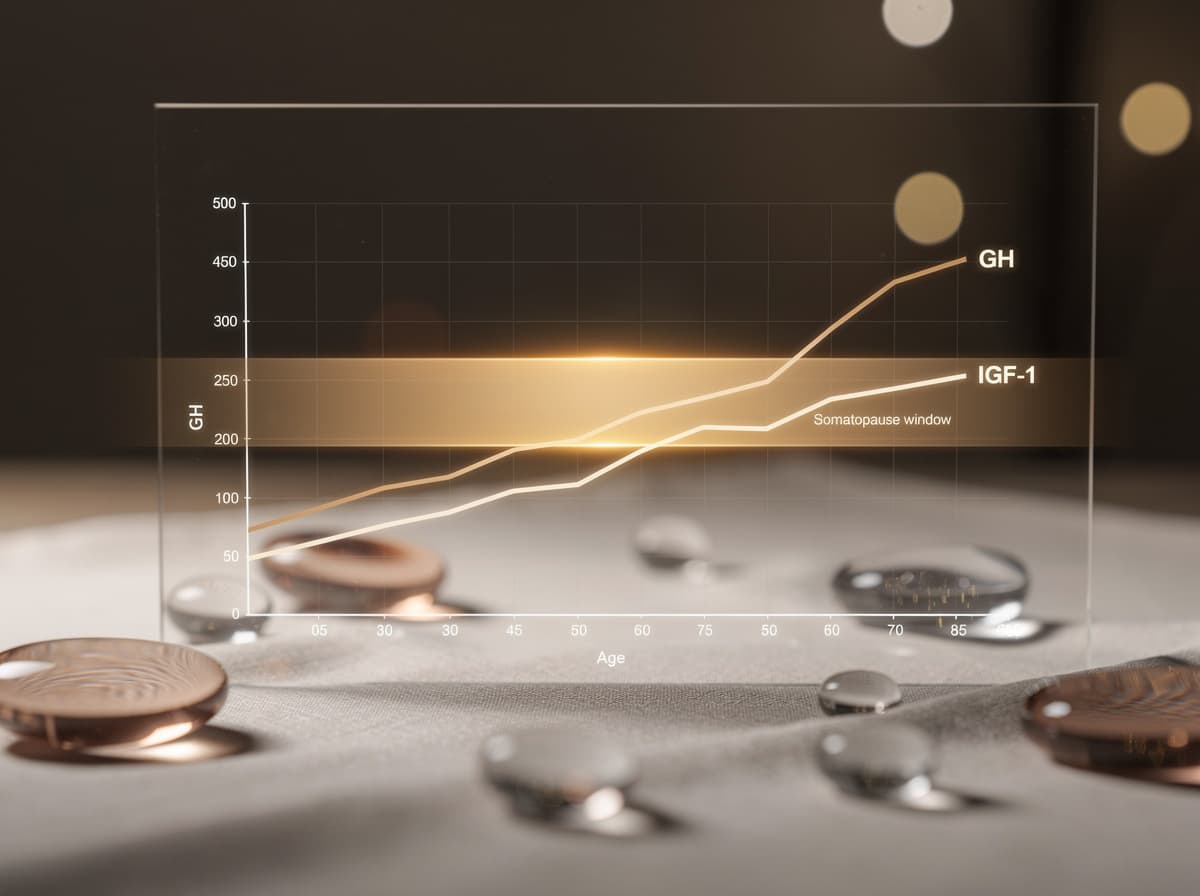
The biological reason interest in GH-axis peptides exists is that the axis attenuates with age. Research has shown that GH production and secretion, GH binding protein, and IGF-1 levels decline with aging, often referred to as the "somatopause", and these decrements provide the basis for speculation that these anabolic hormones are related to body composition, metabolic, and functional changes that occur with aging.
The numbers are starker than most people expect. After puberty, GH levels decrease exponentially, production is reported to decline by 14% per decade and may decline by up to 50% every seven years for adult men, with elderly men producing as little as 50 μg/day compared with pubertal boys who produce 1.0 to 1.5 mg/day. Studies have shown the resulting state is associated with increased adipose tissue and the broader cluster of changes that mirror clinical adult GH deficiency.
A second clinical review frames the mechanism plainly: age-related decline in GH levels is very well documented, consistent across different mammalian species and primarily due to reduced hypothalamic secretion of GHRH with consequent decline of GH biosynthesis and release by the anterior pituitary, which leads to a reduction in circulating levels of insulin-like growth factor 1 (IGF-1), the key mediator of GH action.
Two preliminary findings matter for protocol thinking. First, the decline is upstream: it starts with GHRH, not with somatotroph capacity, which is why GHRH analogs remain effective in older subjects. Second, the tesamorelin literature documents that the GHRH agonist restores normal GH pulsatility and amplitude, selectively reduces visceral fat, intima media thickness and triglycerides, and improves cognitive function in older persons. Preliminary evidence points toward restoring pulse architecture, not flooding the system with continuous GH.
Feedback loops and side-effect mechanisms come from the same diagram#
Reading the axis as a loop also explains why some compounds cause specific tolerability profiles. Three feedback nodes matter:
- IGF-1 negative feedback at the hypothalamus. Rising hepatic IGF-1 inhibits GHRH neurons and stimulates somatostatin. This is why chronic high-dose ghrelin agonism, especially with oral MK-677, can plateau over weeks: the elevated IGF-1 closes the loop and dampens further GH amplitude.
- Ghrelin's orexigenic action. The same receptor that drives GH release also drives hunger. Research has shown that ghrelin is an orexigenic hormone, released primarily by the gastric mucosa, although it is widely expressed in many different tissues, including the central nervous system and the pituitary. Increased appetite on MK-677 is not a side effect; it is on-target pharmacology.
- Insulin and glucose handling. GH is counter-regulatory to insulin. Studies have shown that during prolonged GH elevation, hepatic gluconeogenesis and free fatty acid mobilization rise, which can blunt insulin sensitivity. The peptide calculator integrates this when modelling pulse exposure over a cycle.
A separate point worth flagging: somatostatin inhibits GH secretion and regulates multiple processes by signaling through its receptors sst1 to sst5, and differential expression of these receptors may contribute to sex-specific GH patterns and fasting-induced GH rise. Translation: protocol responses differ by sex and feeding state, and a single dose recommendation across populations is rarely defensible.
How the four GH-axis peptides map onto this circuit#
Putting it together, the four peptides Klarovel covers in depth occupy distinct positions on the same diagram:
- Ipamorelin: short-acting selective GHSR-1a agonist. Produces a clean GH pulse without significant cortisol or prolactin elevation. Best for users who want pulsatile, time-bounded stimulation.
- CJC-1295 (with or without DAC): GHRH receptor agonist. Without DAC, behaves like a short GHRH pulse; with DAC, provides sustained GHRH receptor exposure. Often paired with ipamorelin to engage both receptors.
- MK-677 (ibutamoren): orally bioavailable, long-acting GHSR-1a agonist. Maintains elevated 24-hour GH and IGF-1 averages but loses pulse architecture and engages the orexigenic and water-retention effects of ghrelin signalling more visibly.
- Tesamorelin: stabilised GHRH analog with the clearest clinical evidence base, specifically for visceral adipose tissue reduction in HIV-associated lipodystrophy per its FDA labelling.
For a fuller framework on how to compare these candidates, see how Klarovel structures protocol curation, and run dose-window scenarios through the peptide calculator. Account holders can save protocol templates via register.
Frequently asked questions about the GH peptide axis#
What is the difference between a GHRH analog and a ghrelin mimetic?
They activate different receptors on the same pituitary cells. GHRH analogs (CJC-1295, tesamorelin) bind the GHRH receptor; ghrelin mimetics (ipamorelin, MK-677) bind GHSR-1a. Research has shown the two receptors converge on complementary intracellular pathways, which is why combining them produces a larger pulse than either alone.
Why do protocols often pair two peptides instead of using one at a higher dose?
Because the dose-response curve at a single receptor saturates. The paired approach exploits two independent receptors, each contributing to GH release. Studies have shown that ghrelin acts synergistically with GHRH, stimulates GH secretion independently of GHRH receptors, acts at both the hypothalamic and pituitary levels, and may functionally antagonize somatostatin. Two distinct mechanisms, one outcome.
Does the GH axis stop working with age?
No, but the upstream signal weakens. The somatotrophs in the pituitary remain largely responsive; what declines is hypothalamic GHRH drive and pulse organisation. Preliminary evidence suggests this is why GHRH analogs such as tesamorelin retain efficacy in older subjects.
Is the somatopause a disease?
Not in clinical terms. It is a normal age-related shift. It is associated with changes in lean mass and adiposity that overlap with adult GH-deficiency phenotypes, but consensus guidelines do not classify it as a deficiency state requiring intervention.
Why does MK-677 increase appetite when ipamorelin generally does not?
Both engage GHSR-1a, but their pharmacokinetics differ. MK-677 maintains receptor occupation across 24 hours, which engages ghrelin's orexigenic central effects continuously. Ipamorelin's short half-life produces a discrete pulse that subsides before the appetite circuitry is meaningfully activated.
Where does IGF-1 fit in protocol design?
IGF-1 is the downstream readout of GH-axis activity and the negative-feedback signal back to the hypothalamus. Tracking IGF-1 over a cycle gives a useful proxy for whether a protocol is producing biologically meaningful exposure, while also flagging when the negative feedback loop is closing and pulse amplitude is being suppressed.
The axis view changes which questions are worth asking#
Instead of asking "which peptide is best", a working knowledge of the GH peptide axis reframes the question: which receptor, which pulse pattern, which feedback regime fits the goal. That framing is what Klarovel's protocol curation is built around. Start with the peptide calculator to model a candidate protocol, review the regulatory framing in our disclosures, and register to save your work and follow the cluster updates as new evidence lands.
Keep reading
What is peptide therapy? Three categories that matter
Peptide therapy means three different things in 2026: FDA-approved medicines, compounded peptides in a reopened grey zone, and research-only peptides.

The GLP-1 class explained: how semaglutide, tirzepatide, and retatrutide differ
Semaglutide, tirzepatide, and retatrutide share a pharmacology family but each activates different metabolic receptors. Here's what the difference actually means.
Melanotan 2 in Norway: Law, Risks, and Safer Tan Options
What research shows about Melanotan 2, why DMP has not approved it in Norway, the documented melanoma case reports, and evidence-based alternatives for pigmentation goals.