Retatrutide: What the triple-agonist trials actually show
Retatrutide is the first triple GIP-GLP-1-glucagon agonist. Phase 3 trials show 28.7% weight loss at 68 weeks. Here's what the evidence actually shows.
The retatrutide peptide has produced the largest average weight loss ever reported in an obesity Phase 3 trial: 28.7% in 68 weeks. The molecule is not approved anywhere as of April 2026. Understanding the retatrutide peptide requires holding both facts at once.
Retatrutide targets three metabolic receptors at once#
Retatrutide (Lilly code LY3437943) is a first-in-class triple agonist. It is a synthetic 39-amino-acid peptide engineered from a GIP backbone that activates the receptors for gastric inhibitory polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon in a single molecule. Relative potency differs across the three receptors: retatrutide is more potent at the human GIP receptor than at GLP-1 or glucagon.
The reason to add glucagon to a GLP-1/GIP combination is broader metabolic engagement. Studies have shown GLP-1 suppresses appetite and slows gastric emptying. GIP potentiates insulin secretion and acts on adipose tissue. Glucagon agonism has been shown to raise resting energy expenditure, mobilize hepatic fat, and increase lipolysis. A single weekly subcutaneous dose engages all three pathways.
This is where retatrutide is clinically distinct from the approved GLP-1 medicines. Semaglutide activates one receptor. Tirzepatide activates two. Retatrutide activates three. Each step up the receptor count corresponds to larger average weight-loss effects in head-to-head and cross-trial data.
Phase 2 established the ceiling: 24.2% mean weight loss in 48 weeks#
The Phase 2 obesity trial led by Ania Jastreboff and colleagues (NEJM, 2023, NCT04881760) enrolled 338 adults with obesity. At the 24-week primary endpoint, body-weight changes were -7.2% at 1 mg, -12.9% at 4 mg, -17.3% at 8 mg, and -17.5% at 12 mg, versus -1.6% on placebo. At 48 weeks, the 12 mg dose had produced a mean 24.2% body-weight reduction.
For context, the comparable 48-week to 68-week endpoints for semaglutide 2.4 mg weekly sit near 14.9%, and tirzepatide 15 mg weekly reached approximately 22.5% at 72 weeks in SURMOUNT-1. Retatrutide's Phase 2 numbers were, at the time of publication in 2023, the largest weight reductions documented for any pharmacotherapy in a general obesity population.
The Phase 2 tolerability profile was class-typical: gastrointestinal events that were dose-dependent, mostly mild to moderate, and concentrated in the dose-escalation period. Nausea reached roughly 60% at the 12 mg dose; vomiting, diarrhea, and constipation followed at lower rates.
Phase 3 TRIUMPH-4 confirmed the signal, and introduced a new one#
TRIUMPH-4 was the first Phase 3 readout for retatrutide, announced in December 2025. The trial enrolled adults with obesity and co-occurring knee osteoarthritis, measured body weight and pain at 68 weeks, and compared retatrutide 9 mg and 12 mg to placebo.
The efficacy numbers exceeded the Phase 2 results:
- 12 mg: 28.7% mean body-weight reduction at 68 weeks
- 9 mg: 26.4% mean body-weight reduction at 68 weeks
- 12 mg: 58.6% of participants achieved ≥25% body-weight loss
- Systolic blood pressure: -14.0 mmHg at 12 mg
- Osteoarthritis pain: approximately 75% reduction on both retatrutide doses versus 40% on placebo
The safety profile, however, introduced signals that are worth taking seriously. The Lilly Phase 3 release reported that dysesthesia (altered skin sensation, often described as tingling or burning) occurred in 20.9% of patients at 12 mg versus 0.7% on placebo. The effect is associated with glucagon-receptor activity and has no direct analog in the approved GLP-1 medicines.
Discontinuation because of adverse events was 18.2% at 12 mg, 12.2% at 9 mg, and 4% on placebo. A meta-analysis pooling three retatrutide RCTs (878 patients) found a risk ratio of 6.70 (p = 0.002) for treatment discontinuation at the 12 mg dose relative to placebo.
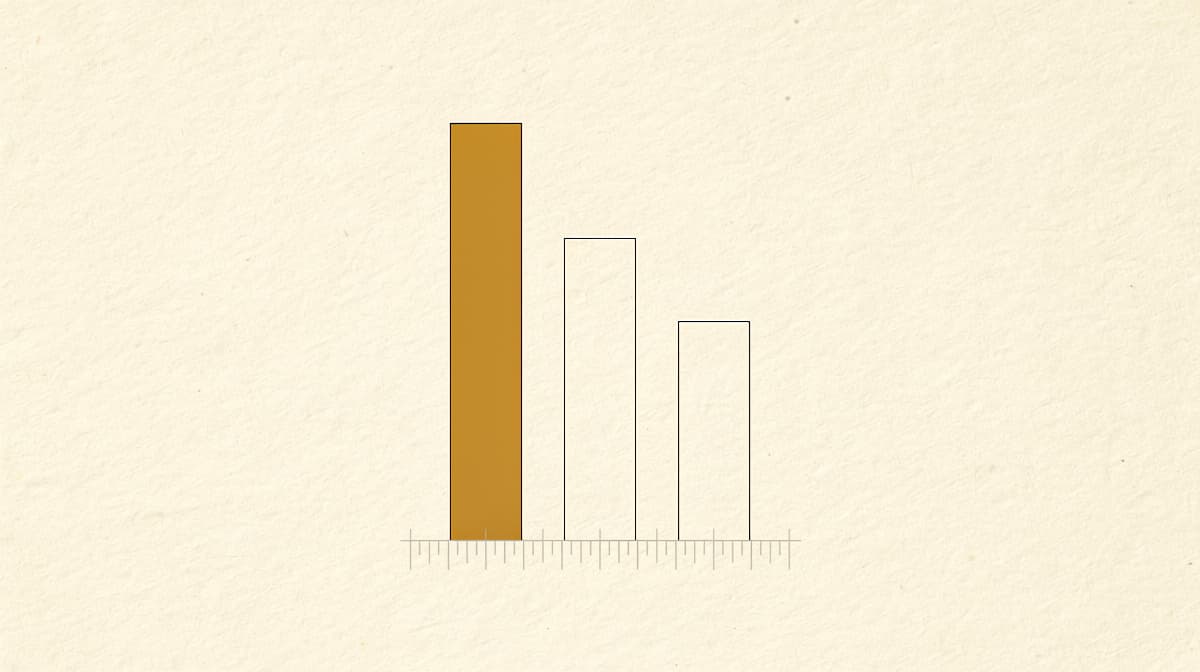
Retatrutide vs tirzepatide vs semaglutide: the efficacy gap is real#
Direct head-to-head trials of the three molecules do not yet exist. Cross-trial comparison is imperfect, but the endpoints are close enough to be informative:
| Drug | Receptor class | Highest-studied dose | Weight loss | Timepoint |
|---|---|---|---|---|
| Semaglutide | GLP-1 | 2.4 mg weekly | 14.9% | 68 wk (STEP 1) |
| Tirzepatide | GIP + GLP-1 | 15 mg weekly | 22.5% | 72 wk (SURMOUNT-1) |
| Retatrutide | GIP + GLP-1 + glucagon | 12 mg weekly | 28.7% | 68 wk (TRIUMPH-4) |
The stepwise pattern tracks with the stepwise addition of receptor engagement. This is the single most interesting clinical-pharmacology story in obesity medicine right now. It is also why glucagon-specific side effects (dysesthesia, higher heart-rate signal, higher discontinuation) need to be evaluated on their own terms rather than assumed to be a straight extension of the GLP-1 class.
Beyond obesity: T2D, MASLD, and cardiovascular signals#
Retatrutide's clinical program extends well past weight loss.
In Phase 2b type-2 diabetes data (Lancet 2023), retatrutide produced HbA1c reductions that compared favorably to active comparators. The pooled meta-analysis of three RCTs reported a mean HbA1c reduction of -0.91% overall and -1.54% at the 12 mg dose, with systolic blood-pressure reductions of -9.88 mmHg and diastolic reductions of -3.88 mmHg.
A Phase 2a trial in metabolic dysfunction-associated steatotic liver disease (MASLD), published in Nature Medicine (2024), showed meaningful reductions in liver fat content at doses used for obesity, a finding that matters because MASLD currently has limited approved pharmacotherapy.
A dedicated cardiovascular outcomes trial is enrolling. Early surrogate-marker data (blood pressure, lipids, liver fat) point in a favorable direction, but the cardiovascular endpoint that insurers and regulators care about will take years.
Retatrutide is not approved anywhere: what that means today#
As of April 2026, retatrutide is not FDA-approved, not authorized by the European Medicines Agency, and not authorized by DMP (the Norwegian Medicines Agency). Eli Lilly has not yet filed a New Drug Application. The first two Phase 3 readouts (TRIUMPH-4 in December 2025 and TRANSCEND-T2D-1 in March 2026) sit inside an ongoing Phase 3 program; the full TRIUMPH and TRANSCEND program is expected to complete in 2026, with regulatory filings following.
In practical terms:
- Legally, retatrutide is investigational. It is not available through pharmacies in the US, EU, or Norway. Any prescription-adjacent product sold under the retatrutide name in those jurisdictions is not an approved medicine.
- Research-grade retatrutide is distributed through specialized suppliers. Klarovel's catalog lists Retatrutide in both vial (10 mg and 30 mg) and pen (12 mg and 40 mg) formats. Anyone considering whether to retatrutide buy a research-grade product should first read the regulatory framing on our disclosures page: research-grade retatrutide is supplied through vetted distributors for research purposes only, not as an approved medicine, and the retatrutide buy decision sits inside that research-only frame rather than a clinical prescribing one.
- Structural oversight matters. Retatrutide's Phase 3 discontinuation rate, its dysesthesia signal, and its activity across three metabolic axes make it a molecule where baseline bloodwork and structured screening are not optional. Klarovel's intake questionnaire screens for the class-level contraindications: pregnancy and breastfeeding, active or recent cancer, uncontrolled hypertension, significant renal or hepatic impairment, and personal or family history of medullary thyroid carcinoma or MEN 2.
Frequently asked questions about retatrutide#
Is retatrutide approved anywhere yet?
No. As of April 2026, retatrutide is not approved by the FDA, the European Medicines Agency, or DMP in Norway. It is in active Phase 3 trials (the TRIUMPH and TRANSCEND programs). Eli Lilly has not yet filed a New Drug Application. Any product sold in approved-pharmacy channels under the retatrutide name in those jurisdictions is not an approved medicine.
How much weight loss does retatrutide produce compared to tirzepatide and semaglutide?
In Phase 2 trials, retatrutide produced 24.2% mean weight loss at 48 weeks at the 12 mg dose. Tirzepatide produced approximately 22.5% over 72 weeks in SURMOUNT-1, and semaglutide produced approximately 14.9% over 68 weeks in STEP 1. The efficacy gap between retatrutide and the dual or single agonists is real and reproducible across trials.
Why is retatrutide more effective than dual GLP-1/GIP agonists?
Retatrutide adds glucagon-receptor agonism to GLP-1 and GIP. Glucagon-receptor signalling drives lipolysis and increases energy expenditure, an axis the dual agonists do not engage. The triple-agonist mechanism produces additional fat oxidation and resting energy use on top of the appetite-suppression and insulin-sensitivity effects shared with the dual agonists.
What are the most common retatrutide side effects in trials?
The Phase 3 adverse-event profile centres on three findings. Gastrointestinal events (nausea, vomiting, diarrhoea) are the most frequent and lead to discontinuation in close to one in five patients at the 12 mg dose. Dysesthesia (abnormal sensation in extremities) appeared in roughly one in five patients at 12 mg, a signal not observed in the approved GLP-1 comparators. A heart-rate increase not seen with semaglutide or tirzepatide also persisted across trials.
When will retatrutide be approved?
The full TRIUMPH and TRANSCEND programs are expected to complete in 2026, with regulatory filings to follow. A best-case approval window is 2027 to 2028 in the United States; European Medicines Agency and DMP authorisation typically lag United States approval by 6 to 18 months. Cardiovascular outcomes data, due in this window, will shape both the indication and the prescribing audience.
Can I get a retatrutide prescription in Norway today?
No. DMP has not authorised retatrutide. It cannot be dispensed through Norwegian pharmacies, and it is not eligible for legal personal-import under the medicinal-products regulation. For research-grade retatrutide, distribution runs through specialised research suppliers such as the network listed in the Klarovel catalogue, with the regulatory caveats explained on our disclosures page.
What to watch in the next twelve months#
Five data points will shape the retatrutide story through 2026 and into 2027: the TRIUMPH-1 general-obesity readout (the largest Phase 3 trial in the program); the TRANSCEND-T2D full efficacy data; the cardiovascular outcomes trial interim analysis; extended safety follow-up on dysesthesia and heart rate; and any regulatory-filing announcement from Lilly.
The early returns read like a new ceiling for pharmacological weight loss. The open questions are long-term tolerability, the real-world durability of effect after discontinuation, and whether the regulatory picture moves from investigational to prescription by 2027 or 2028. For now, retatrutide sits at the edge of approved medicine: promising, investigational, and not the right fit for everyone.
Anyone genuinely considering a retatrutide protocol should start with a structured health profile and recent bloodwork, not with a product SKU. Start with the Klarovel questionnaire to see whether the molecule fits the profile at all. If it does, the product catalog and the reconstitution calculator are the next stops.
Keep reading

What Are Peptides Used For? An Evidence-Based Primer
A clear, research-grounded look at what peptides are used for in 2026: from approved indications like diabetes and obesity to recovery research.

Peptides for Healing: Research-Backed Tissue Repair Guide
How peptides for healing actually work. Mechanisms, evidence, and protocol logic for BPC-157, TB-500, GHK-Cu, and KPV across tendon, skin, and gut.
AOD-9604 peptide: complete guide to the fat-metabolism fragment
AOD-9604 is a 16-amino-acid growth hormone fragment studied for fat metabolism. What the trials actually showed, what they did not, and where it fits today.