Cagrilintide Complete Guide: Amylin Analog for Weight Loss
Cagrilintide is the long-acting amylin analog behind CagriSema. Mechanism, REDEFINE 1 data, dosing, side effects, and comparison to semaglutide, in plain English.

Cagrilintide sits at the center of the next obesity-pharmacology wave: a once-weekly amylin analog peptide that, when paired with semaglutide as CagriSema, delivered the largest weight loss ever recorded in a phase 3 obesity trial. This guide unpacks the cagrilintide mechanism of action, the REDEFINE 1 trial numbers, the cagrilintide dosing protocol used in studies, the cagrilintide side effects readers should expect, and how cagrilintide vs semaglutide actually compares head to head.
Key takeaways#
- Cagrilintide is a long-acting acylated analog of amylin, the satiety hormone co-secreted with insulin from pancreatic beta cells. Its lipid modifications extend its half-life to roughly 160 to 195 hours, supporting once-weekly subcutaneous dosing.
- In the REDEFINE 1 phase 3 trial (n=3,417), the CagriSema combination (cagrilintide 2.4 mg + semaglutide 2.4 mg) produced a mean weight reduction of 20.4% at week 68, versus 3.0% on placebo, with 60% of participants reaching at least 20% weight loss.
- As monotherapy at 4.5 mg, cagrilintide produced roughly 10.8% weight loss at 26 weeks in the Lau et al. phase 2 trial, outperforming liraglutide 3.0 mg (9.0%).
- Cagrilintide is investigational. Novo Nordisk filed an NDA with the FDA for CagriSema in December 2025, with approval realistically targeted for late 2026 or 2027.
- Cagrilintide is not a Klarovel product. Klarovel publishes protocol-layer guidance; research-grade peptides are available from specialised suppliers operating under their own regulatory framing.
Cagrilintide is a re-engineered amylin built for weekly dosing#
Native amylin is a 37-amino-acid peptide that the pancreas releases alongside insulin every time a meal hits. It tells the brain you are full, slows gastric emptying, and suppresses postprandial glucagon. The problem: native amylin has a half-life measured in minutes, and pramlintide, the first synthetic version, requires multiple daily injections and tends to aggregate in solution. Cagrilintide solves both problems through targeted re-engineering.
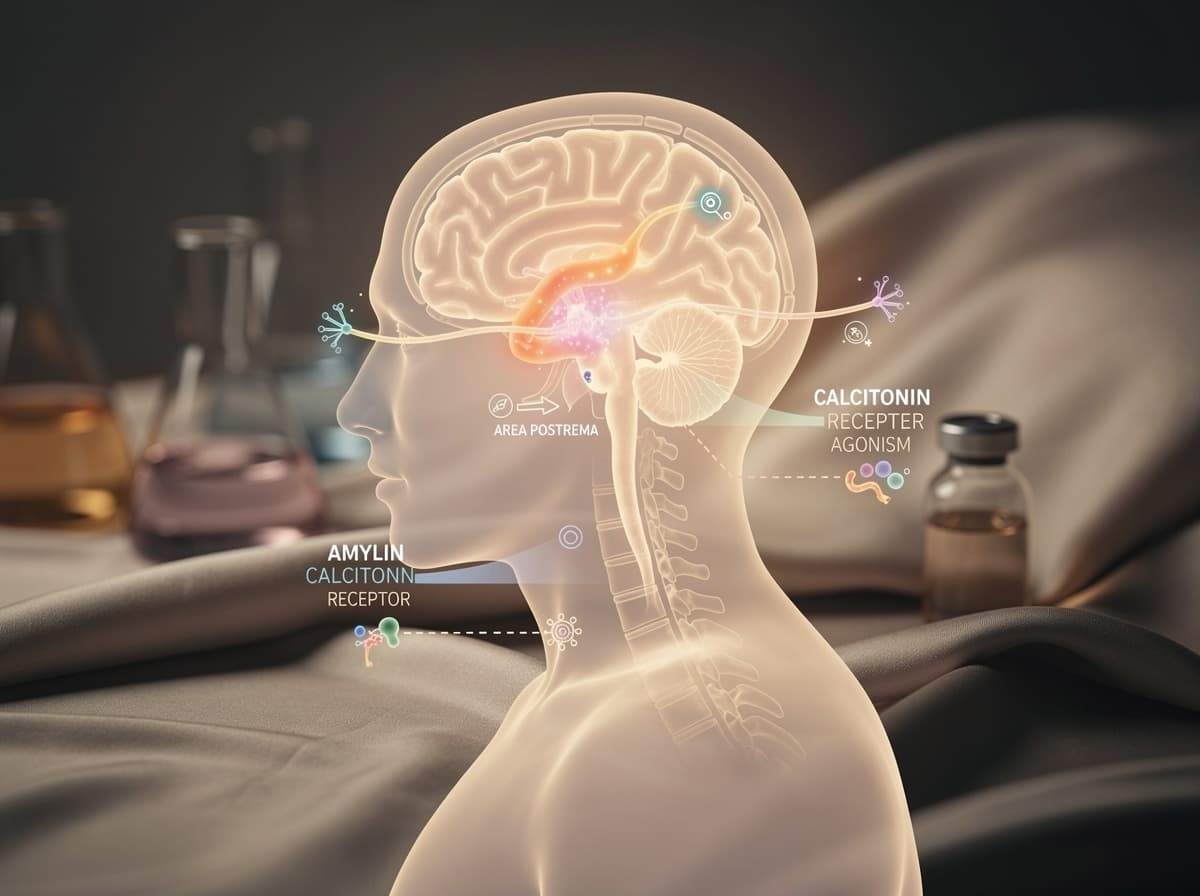
Cagrilintide (development codes AM833 / NN9838) is a peptide engineered by Novo Nordisk aimed at addressing obesity and type 2 diabetes. It is a modified form of amylin, the 37-amino-acid hormone co-released with insulin by the pancreas, which delays gastric emptying, regulates postprandial glucagon secretion, and enhances feelings of fullness . Crucially, cagrilintide acts as a dual amylin and calcitonin receptor agonist (DACRA), engaging both amylin and calcitonin receptors , which broadens its central appetite-suppression footprint.
The cagrilintide mechanism of action depends on two engineering tricks. First, a fatty-acid side chain (lipidation) lets the molecule bind reversibly to albumin in circulation, which is the same trick that gives semaglutide its weekly dosing window. Second, residue substitutions stabilise the peptide against aggregation. The net result: lipid modifications extend its half-life to approximately 160 to 195 hours, enabling once-weekly dosing . The amylin analog peptide hits central amylin and calcitonin receptors in the area postrema and hindbrain, producing satiety through pathways completely independent of GLP-1 signalling, which is exactly why pairing it with a GLP-1 agonist is so interesting.
CagriSema weight loss in REDEFINE 1 set a phase 3 benchmark#
The REDEFINE 1 trial is the headline data set. Published in The New England Journal of Medicine in June 2025, it was the first phase 3 readout for cagrilintide combined with semaglutide.
The design was deliberate. In a phase 3a, 68-week, multicenter, double-blind, placebo-controlled and active-controlled trial, adults without diabetes who had a BMI of 30 or higher (or 27 or higher with at least one obesity-related complication) were enrolled. Participants were randomly assigned in a ratio of 21:3:3:7 to receive the combination of semaglutide 2.4 mg and cagrilintide 2.4 mg, semaglutide alone at 2.4 mg, cagrilintide alone at 2.4 mg, or placebo, plus lifestyle interventions for all groups.
The four-arm structure is what makes REDEFINE 1 useful. Every other major obesity phase 3 trial in the past decade has compared a single agent to placebo. REDEFINE 1 lets you read both monotherapies and the combination in the same population on the same timeline.
The numbers, per the Garvey et al. NEJM publication:
Average change in body weight from baseline to 68 weeks: -20.4% with the combination of semaglutide and cagrilintide, -14.9% with semaglutide alone, -11.5% with cagrilintide alone, and -3.0% with placebo.
In the REDEFINE 1 trial, 60% of participants receiving once-weekly CagriSema achieved at least 20% weight loss, and 23% lost 30% or more, which is among the most significant results seen with any anti-obesity medication to date.
The combination therapy improved systolic blood pressure, waist circumference, lipid levels, and glycemic control, with 88% of participants with prediabetes returning to normoglycemia.
The CagriSema weight loss signal in REDEFINE 1 also has a higher-adherence number worth knowing. When accounting for full treatment adherence, weight loss with CagriSema rose to 22.7%, with 40.4% of participants achieving at least 25% weight loss. That is the trial-product estimand, which models what happens if participants stay on study drug for the full 68 weeks. The 20.4% figure is the treatment-policy estimand, which keeps participants in the analysis even after discontinuation, and is closer to a real-world adherence picture.
For context on cagrilintide monotherapy, the phase 2 data is still relevant because it established the 4.5 mg ceiling and the liraglutide comparison. In Lau et al. Lancet 2021, adults with overweight or obesity (BMI 27-40 kg/m²) were randomized to one of seven cagrilintide doses (0.3 to 4.5 mg weekly), liraglutide 3.0 mg daily as an active comparator, or placebo, all for 26 weeks. At the highest dose (4.5 mg), cagrilintide produced 10.8% weight loss at 26 weeks versus 3.0% with placebo and 9.0% with liraglutide 3.0 mg. Worth emphasising: cagrilintide 4.5 mg as monotherapy beat liraglutide 3.0 mg, the first-generation daily GLP-1 agonist. Amylin receptor agonism on its own, with no incretin involvement, produces results competitive with first-generation GLP-1 therapy.

Cagrilintide dosing follows a 16-week titration to 2.4 mg weekly#
Every published efficacy signal for the combination anchors to a 2.4 mg weekly target dose, escalated gradually. The phase 2 monotherapy dose-response showed that higher doses produced more weight loss but also more nausea, which is why titration exists. The phase 3 REDEFINE trials and the pending NDA both use 2.4 mg as the steady-state target, mirroring semaglutide's Wegovy schedule.
Research suggests the titration protocol used in REDEFINE 1 looked like this:
- Escalation phase (16 weeks): stepwise increases from 0.25 mg to 2.4 mg weekly, doubling roughly every 4 weeks. This is the same shape as semaglutide titration.
- Maintenance phase: 2.4 mg once weekly subcutaneous.
- If stacking with semaglutide: the REDEFINE protocol co-administers each at 2.4 mg, with parallel titration. Both semaglutide and cagrilintide were titrated up from initial doses of 0.25 mg.
In the phase 2 monotherapy trial, the higher 4.5 mg ceiling produced incremental weight loss over 2.4 mg, but the phase 3 program standardised on 2.4 mg because that is the dose that pairs cleanly with semaglutide 2.4 mg in the CagriSema fixed-dose product. The cagrilintide dosing rule of thumb: titrate slowly, hold the dose if nausea is significant, and do not chase a higher dose to compensate for a stalled scale. Preliminary evidence points to most of the GI burden landing in the escalation window, not at steady state.
Cagrilintide side effects are mostly gastrointestinal and front-loaded#
The cagrilintide side effects profile, both as monotherapy and combined with semaglutide, is dominated by transient gastrointestinal events. This is consistent across all published trials.
In the phase 2 monotherapy trial, the most frequent adverse events were gastrointestinal disorders (e.g. nausea, constipation, and diarrhoea) and administration-site reactions. More participants receiving cagrilintide 0.3 to 4.5 mg had gastrointestinal adverse events compared with placebo (41% to 63% vs 32%), primarily nausea (20% to 47% vs 18%). The dose-dependence is the headline: higher doses, more nausea, which is why the titration protocol exists.
In REDEFINE 1, the combination side-effect profile was meaningfully heavier than either monotherapy. Gastrointestinal adverse events (affecting 79.6% in the cagrilintide-semaglutide group and 39.9% in the placebo group), including nausea, vomiting, diarrhea, constipation, or abdominal pain, were mainly transient and mild-to-moderate in severity. Discontinuation rates tell a calmer story: discontinuations due to adverse events occurred among 5.9% of the combined treatment group compared with 2.6% to 3.6% in the other treatment groups , which suggests most people pushed through the GI window.
A few things to flag on cagrilintide side effects that are not raw GI:
- Cagrilintide's calcitonin receptor activity raises a theoretical question about bone metabolism, though no bone-related adverse signal has emerged in the trials so far.
- Research has shown injection site reactions occur at modestly higher rates with the combination, in part because (in non-fixed-dose protocols) two separate injections are required.
- Cagrilintide monotherapy has been associated with lower vomiting rates than semaglutide. This tolerability advantage is one reason Novo Nordisk is also pursuing standalone cagrilintide through the RENEW phase 3 programme.
For complete trial safety appendices, see the REDEFINE 1 supplementary materials at NEJM.
Cagrilintide vs semaglutide: complementary, not competitive#
The cagrilintide vs semaglutide question is the most common one readers bring to this molecule, and the answer is structural: they are not competitors. They are complementary mechanisms designed to be stacked.
Mechanism. Semaglutide is a GLP-1 receptor agonist. It works mainly through incretin pathways: slowing gastric emptying, suppressing glucagon, and acting on hypothalamic GLP-1 receptors to reduce hunger. Cagrilintide is a dual amylin and calcitonin receptor agonist. It acts on the area postrema, the hindbrain, and the hypothalamus through amylin and calcitonin pathways. The two pathways converge on appetite suppression but enter the brain through different doors.
Effect size in cagrilintide vs semaglutide head-to-head. REDEFINE 1 ran them in the same trial. Monotherapy semaglutide produced 14.9% weight loss at 68 weeks. Monotherapy cagrilintide produced 11.5%. The combination produced 20.4%. The combination is supra-additive versus either alone, which the trial authors attribute to the complementary effect of the two molecules on appetite regulation through direct actions in brain regions known to be involved in hedonic and homeostatic appetite regulation, such as the hypothalamus, hindbrain, and septum .
Tolerability in cagrilintide vs semaglutide comparisons. Cagrilintide monotherapy generally has a milder GI footprint than semaglutide. The combination has a heavier one than either alone. This matters for protocol design: if a patient cannot tolerate semaglutide at all, cagrilintide monotherapy is a different option, not a similar one.
Regulatory status. Semaglutide is approved (as Wegovy for obesity, Ozempic for type 2 diabetes). Cagrilintide is not approved as monotherapy. CagriSema is the lead asset: Novo Nordisk filed its New Drug Application with the FDA for CagriSema on December 18, 2025. The FDA review is expected through 2026, with approval possible in late 2026 or 2027. If approved, CagriSema would be the first combination obesity drug targeting both amylin and GLP-1 pathways.
The strategic read: cagrilintide is not "the next semaglutide". It is the molecule that makes the next-generation combination work. That distinction matters when planning a stack.
Frequently asked questions about cagrilintide#
Is cagrilintide the same thing as CagriSema?
No. Cagrilintide is the single molecule, a long-acting amylin and calcitonin receptor agonist. CagriSema is the fixed-dose combination product that pairs cagrilintide 2.4 mg with semaglutide 2.4 mg in one weekly injection. CagriSema is a fixed-dose combination of a long-acting amylin analogue, cagrilintide 2.4 mg, and semaglutide 2.4 mg. Cagrilintide can in principle be used as monotherapy (Novo Nordisk is running the RENEW phase 3 program for that indication), but CagriSema is the lead clinical asset and the larger commercial bet.
Is cagrilintide FDA approved?
Not as of May 2026. Cagrilintide is investigational. The closest approval path is CagriSema, for which Novo Nordisk filed its NDA in December 2025. FDA review is ongoing, with approval realistically possible in late 2026 or 2027. Standalone cagrilintide is still in the RENEW phase 3 program. Until approval, all cagrilintide use outside a clinical trial sits in the research-chemical space, which is why availability runs through specialised suppliers operating under their own regulatory framing rather than pharmacies.
How much weight loss should a person expect on cagrilintide monotherapy?
Studies have shown roughly 10% body weight reduction at 26 weeks with 2.4 mg monotherapy and roughly 10.8% with 4.5 mg, both in adults with overweight or obesity. In the REDEFINE 1 phase 3 arm, cagrilintide 2.4 mg alone produced 11.5% weight loss at 68 weeks. The combination with semaglutide roughly doubles those numbers. Real-world responses vary significantly, and weight is generally regained after discontinuation, as is the pattern with all GLP-1 class agents.
What are the most common cagrilintide side effects?
Nausea, vomiting, diarrhea, constipation, and abdominal pain, all gastrointestinal, all dose-dependent, and mostly concentrated in the first 16 weeks of titration. Injection site reactions are the second category. The vast majority of GI events are mild to moderate and transient. Discontinuation rates due to adverse events in REDEFINE 1 stayed under 6% even for the combination, which suggests the side-effect window is generally survivable with a slow titration.
Does cagrilintide cause more or less nausea than semaglutide?
As monotherapy, cagrilintide is associated with somewhat less nausea and vomiting than semaglutide at comparable doses. In combination as CagriSema, gastrointestinal events are more frequent than with either monotherapy alone, because the two mechanisms each contribute their own GI burden. The combined rate in REDEFINE 1 was 79.6% versus 39.9% on placebo, but most events were mild to moderate.
Can cagrilintide be stacked with tirzepatide instead of semaglutide?
No human clinical data exists for cagrilintide plus tirzepatide. The published combination data is exclusively with semaglutide (CagriSema). Tirzepatide is held by a competing sponsor (Eli Lilly), and no formal trial has been announced. Stacking cagrilintide with tirzepatide is community-level speculation, not a research-supported protocol. The Klarovel position: if the data is not in a peer-reviewed trial, the protocol is not ready.
What this means for the GLP-1 stack#
Cagrilintide is the molecule that breaks the single-receptor ceiling. For roughly a decade, GLP-1 receptor agonism has carried the entire weight-management pharmacology load, first as liraglutide, then semaglutide, then tirzepatide as a GLP-1/GIP dual agonist. CagriSema is the first phase 3 demonstration that adding a non-incretin pathway, amylin, produces clearly supra-additive results rather than a marginal lift. The amylin analog peptide is doing real work here, not just smoothing semaglutide's edges.
For readers building a personal protocol library, this matters in three ways. First, the cagrilintide dosing schedule is mature: 2.4 mg weekly after a 16-week titration is the consensus target, validated across two phase 3 trials. Second, the cagrilintide vs semaglutide question is no longer either/or; it is how the two combine. Third, the cagrilintide side effects burden is manageable, but front-loaded, so titration discipline is the single biggest determinant of whether a person stays on protocol through the GI window.
Klarovel publishes protocol-layer guidance for cagrilintide and the broader GLP-1 stack. To access the full protocol library, dose calculators, and the peptide calculator, create a free Klarovel account. For the editorial framing behind our coverage, see how Klarovel works and our disclosures. For adjacent reading, the semaglutide protocol guide covers the GLP-1 side of the same stack.
Cagrilintide is not a fad and not a finished product. It is the next piece of the obesity-pharmacology architecture, and the readers who understand it now will be the ones reading next year's regulatory headlines instead of being surprised by them.
Keep reading

What Are Peptides Used For? An Evidence-Based Primer
A clear, research-grounded look at what peptides are used for in 2026: from approved indications like diabetes and obesity to recovery research.

Peptides for Healing: Research-Backed Tissue Repair Guide
How peptides for healing actually work. Mechanisms, evidence, and protocol logic for BPC-157, TB-500, GHK-Cu, and KPV across tendon, skin, and gut.
AOD-9604 peptide: complete guide to the fat-metabolism fragment
AOD-9604 is a 16-amino-acid growth hormone fragment studied for fat metabolism. What the trials actually showed, what they did not, and where it fits today.