Ipamorelin vs CJC-1295: GHRP vs GHRH Head-to-Head (2026)
Ipamorelin vs CJC-1295 settled: one is a ghrelin-receptor pulse, the other a GHRH analog with a six-day half-life. Here is which to pick, and when.

Researchers comparing growth hormone secretagogues almost always land on the same fork in the road: ipamorelin or CJC-1295. They sound interchangeable in the marketing copy, but they sit on two completely different receptors, follow two completely different pharmacokinetic curves, and answer two completely different research questions. This guide resolves the confusion with primary literature, then gives a decision rule.
Key takeaways#
- Ipamorelin is a GHRP (ghrelin receptor agonist) with a short pulse; CJC-1295 is a GHRH analog, and the DAC version has a half-life measured in days, not minutes.
- Ipamorelin's selectivity is its single biggest differentiator: even at doses more than 200-fold above the ED50 for GH release, it does not raise ACTH or cortisol meaningfully (Raun et al. 1998).
- A single subcutaneous CJC-1295 DAC injection has been shown to elevate plasma GH 2- to 10-fold for six days or longer, and IGF-1 by 0.5- to 3-fold for nine to eleven days (Teichman et al. 2006).
- The two are mechanistically complementary, not competitive. The most-studied combination in the GH-axis literature stacks a GHRH analog with a GHRP.
- For most research protocols asking "which one first?", ipamorelin is the cleaner starting point. CJC-1295 with DAC is the right pick when sustained IGF-1 elevation is the endpoint.
How ipamorelin works#
Ipamorelin is a synthetic pentapeptide with the sequence Aib-His-D-2-Nal-D-Phe-Lys-NH2, characterised in the original Raun et al. work as a high-potency GH releaser both in vitro and in vivo. It binds the growth hormone secretagogue receptor (GHS-R1a), the same receptor that endogenous ghrelin activates in the anterior pituitary. The result is a fast, pulsatile GH release that mimics the natural rhythm.
What separates ipamorelin from earlier GHRPs is what it does not do. None of the secretagogues tested in the Raun work affected FSH, LH, PRL, or TSH plasma levels, but both GHRP-6 and GHRP-2 elevated ACTH and cortisol meaningfully. Ipamorelin did neither, even at doses well above its ED50 for GH release. Research suggests this clean selectivity is why ipamorelin remains the reference GHRP in modern comparison studies, twenty-plus years after its discovery at Novo Nordisk in 1998.
Half-life is short. Studies have shown a Tmax of roughly 10 minutes in animal models, with the GH pulse resolving within an hour or two. That short window is a feature, not a bug, when researchers want to isolate a single GH pulse from background.
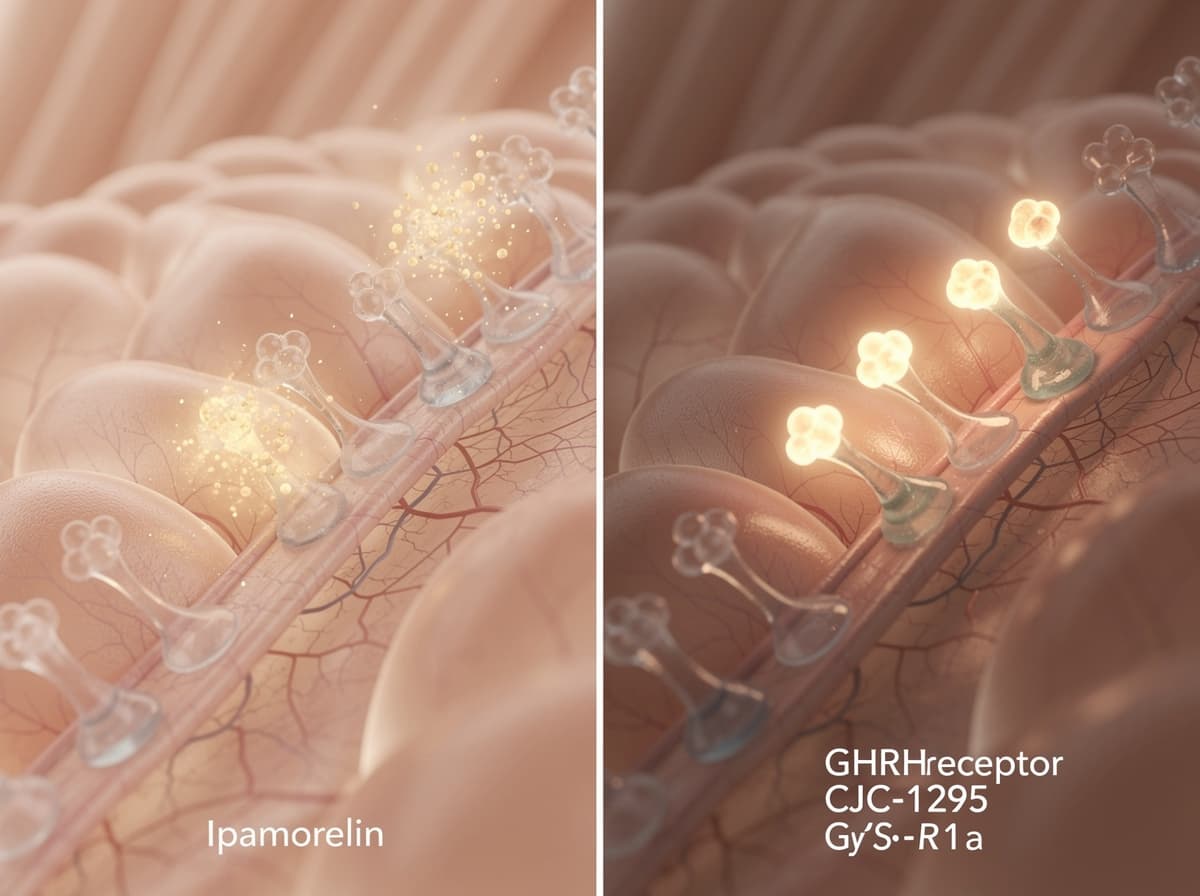
How CJC-1295 works#
CJC-1295 is a modified GHRH analog. Four amino-acid substitutions make the core peptide resistant to dipeptidyl peptidase IV (DPP-IV) cleavage, and a drug affinity complex (DAC) linker lets the molecule covalently bind endogenous albumin after injection, extending its half-life to a duration similar to albumin's. In other words: it tells the pituitary the same thing GHRH does, but it sticks around for days instead of minutes.
There are two CJC-1295 variants and they are not equivalent. CJC-1295 without DAC (also known as Mod GRF 1-29) preserves the first 29 amino acids of native GHRH but omits the albumin-binding moiety, which collapses the half-life and restores a more pulsatile release pattern closer to native GHRH. Studies have shown the DAC variant produces a sustained "GH bleed" while the no-DAC variant produces discrete pulses.
The half-life data is striking. The Teichman et al. 2006 trial in JCEM estimated a CJC-1295 (DAC) half-life of roughly 6 to 8 days in humans, with a cumulative effect observed after multiple doses. Compare that to ipamorelin's minutes-long window and the divergence becomes obvious.
Dosing: ipamorelin vs CJC-1295#
Research-published dosing differs by an order of magnitude in both amount and frequency, because the pharmacokinetics differ by an order of magnitude.
Ipamorelin: research protocols in the published literature have used subcutaneous doses in the low-microgram-per-kilogram range, typically delivered one to three times daily to capture multiple GH pulses across a 24-hour window. The short half-life means daily dosing is the floor, not the ceiling.
CJC-1295 with DAC: research has shown sustained, dose-dependent increases in GH and IGF-I in healthy adults, with the Teichman trial reporting the compound was safe and relatively well tolerated under ascending single doses and weekly or biweekly schedules. Once-weekly dosing is the standard cadence for the DAC variant because of the multi-day albumin-tethered half-life.
CJC-1295 without DAC: dosing cadence resembles ipamorelin (multiple subcutaneous administrations per day) because, without the DAC moiety, the half-life collapses to roughly 30 minutes.
Fasting state matters for both. Preliminary evidence suggests blood glucose above approximately 90 mg/dL blunts the GH response to either peptide, which is why research protocols cluster the injection into a fasted morning window or a pre-sleep window three or more hours after the last meal. Always confirm exact protocol doses through the Klarovel calculator and the research positioning notes on /disclosures; Klarovel does not stock peptides, and research-grade material is available from specialised suppliers.
Evidence: what the studies actually show#
There is no published head-to-head RCT pitting ipamorelin against CJC-1295 in the same patient cohort. The strongest evidence is the parallel monotherapy literature plus the combination data.
For ipamorelin, the foundational paper is Raun et al. 1998 in European Journal of Endocrinology: the first GHRP-receptor agonist with a selectivity for GH release similar to native GHRH. In conscious swine, ipamorelin released GH with an ED50 of roughly 2.3 nmol/kg and an Emax of approximately 65 ng GH/mL plasma, the safety-and-specificity profile that has anchored every comparison study since.
For CJC-1295, the canonical human trial is Teichman et al. 2006. After a single injection, plasma GH concentrations rose 2- to 10-fold for six days or longer, and IGF-1 concentrations rose roughly 1.5- to 3-fold for nine to eleven days. The estimated CJC-1295 half-life was 5.8 to 8.1 days, and after multiple doses, mean IGF-1 levels remained above baseline for up to 28 days. Pulsatility is preserved despite the sustained tonic GHRH signal: research has shown pulsatile GH secretion persists during continuous stimulation by CJC-1295.
The mechanistic rationale for combining the two is straightforward. Ipamorelin activates GHS-R1a; CJC-1295 activates the GHRH receptor (GHRHR). Published research demonstrates that co-administering a GHS-R1a agonist with a GHRH receptor agonist produces supra-additive GH release compared with either compound alone. Two receptors, one pituitary cell, larger pulse.
Side effects and contraindication profile#
Ipamorelin's safety signal is among the cleanest in the GH secretagogue class, which is downstream of its selectivity. It is associated with minimal cortisol, prolactin, FSH, LH, or TSH disturbance, even at supraphysiological doses. The most commonly reported research-protocol observations are transient injection-site reactions and mild head-rush sensations within minutes of administration, attributable to the ghrelin-receptor activation.
CJC-1295's profile is more mixed. The Teichman trial reported the compound was generally well tolerated, but the long half-life carries downstream considerations. Sustained IGF-1 elevation over weeks is the explicit pharmacodynamic effect, and preliminary evidence suggests this is the same axis implicated in insulin resistance and water retention at higher cumulative exposures. The clinical program was complicated: CJC-1295 reached phase II clinical trials but was discontinued after the death of one trial subject. The attending physician concluded the most likely explanation was asymptomatic coronary artery disease with plaque rupture and occlusion, unrelated to treatment with CJC-1295. Researchers reviewing the CJC-1295 file should read that disclosure in full before designing a long-duration protocol.
Overlap zone: both peptides can produce mild water retention, tingling/paresthesia at the injection site, and short-lived flushing. Both are associated with elevated IGF-1, which is the desired endpoint but also the variable to monitor.
When to choose ipamorelin#
Pick ipamorelin when the research question is any of the following:
- Isolating a single GH pulse without confounders from cortisol, prolactin, or appetite axis activation. Ipamorelin's selectivity profile is the cleanest in the class.
- Pulse-pattern fidelity matters more than total AUC. If the goal is to study GH pulse architecture as it occurs physiologically (4 to 6 pulses per 24 hours, dominated by the slow-wave-sleep pulse), a short-acting GHRP fits the experimental design.
- Naive subjects or first-pass tolerance assessment. Ipamorelin's two-decade safety record and the absence of meaningful HPA-axis activation make it the conservative starting point.
- Female research subjects or aging cohorts where cortisol cross-reactivity from older GHRPs is a known confounder. Studies have shown ipamorelin avoids that issue.

When to choose CJC-1295#
Pick CJC-1295 when the research question is any of the following:
- Sustained IGF-1 elevation is the endpoint. The DAC variant's multi-day half-life produces the cumulative IGF-1 plateau that short-acting GHRPs cannot achieve.
- Adherence-limited protocols. Once-weekly subcutaneous administration vs. multiple daily injections is a real-world adherence delta. CJC-1295 DAC has been shown to maintain elevated GH/IGF-1 for days from a single dose.
- Tonic-vs-pulsatile mechanism research. If the experimental design specifically tests whether continuous GHRH-receptor stimulation preserves pulsatility, CJC-1295 is the only published tool that does this in humans.
- GHRH-axis insufficiency models. In hypothalamic/pituitary research where the GHRH signal itself is the deficit, a GHRH analog answers the question, not a GHRP.
If pulsatile fidelity is the priority but DAC is not desired, the no-DAC variant (Mod GRF 1-29) bridges the two profiles.
Can you stack ipamorelin and CJC-1295?#
Yes, and this is the most-studied GH-axis combination in the secretagogue literature. The biological rationale is straightforward: the two compounds hit different receptors on the same somatotroph cell, and research has shown the resulting GH release is supra-additive rather than merely additive.
In a stack, CJC-1295 (typically the no-DAC variant for protocols seeking pulse fidelity, or the DAC variant for protocols seeking sustained elevation) provides the GHRH-receptor "amplitude" signal, while ipamorelin provides the GHS-R1a "frequency" signal via ghrelin mimicry. The pairing is the most co-searched combination in the GH-axis category, which itself reflects how thoroughly it has saturated the research-protocol literature.
Practical caveat: stacking compounds in research subjects amplifies both the GH/IGF-1 signal and the monitoring burden. IGF-1, fasting glucose, and HbA1c should all sit in the monitoring panel.
Verdict#
For most research protocols asking "which one should I start with?", ipamorelin is the better entry point. Its selectivity profile makes it the cleanest tool to characterise a GH response without cortisol, prolactin, or appetite confounders, and its short half-life keeps the experimental window tight. The 1998 Raun paper plus twenty-plus years of safety follow-up gives ipamorelin the strongest evidentiary base in the GHRP class.
CJC-1295 wins on a narrower but real terrain: when the endpoint is sustained IGF-1 elevation over days, when adherence demands once-weekly dosing, or when GHRH-receptor pharmacology is itself the variable under study. The DAC variant's 6-to-8-day half-life is unique in the class and changes what kind of protocol is even possible.
The stack is what most experienced researchers actually run, and the data supports that choice. But if forced to pick one as a starting tool, ipamorelin's signal-to-noise ratio is hard to beat.
Frequently asked questions about ipamorelin vs CJC-1295#
Is CJC-1295 stronger than ipamorelin?
"Stronger" depends on the metric. CJC-1295 produces a larger and longer cumulative GH/IGF-1 elevation per dose, particularly the DAC variant, which has been shown to elevate plasma GH 2-to-10-fold for six days or more. Ipamorelin produces a sharper, shorter, more physiological pulse. Research suggests neither dominates the other on peak GH amplitude in a single pulse; CJC-1295 dominates on AUC, ipamorelin dominates on pulse fidelity.
Are ipamorelin and CJC-1295 the same thing?
No, and this is the most common confusion in the category. Ipamorelin is a GHRP that activates the ghrelin receptor (GHS-R1a). CJC-1295 is a GHRH analog that activates the GHRH receptor (GHRHR). They are mechanistically complementary, which is why they are so commonly stacked, but they are not interchangeable.
Does ipamorelin raise cortisol like other GHRPs?
No. This is its defining feature. The 1998 Raun paper documented that ipamorelin produced no significant ACTH or cortisol elevation even at doses 200-fold above the GH-releasing ED50, in sharp contrast to GHRP-2 and GHRP-6 which both elevated cortisol meaningfully.
What is the difference between CJC-1295 with DAC and without DAC?
The DAC (drug affinity complex) is a small chemical addition that covalently binds the peptide to endogenous albumin after injection. With DAC, the half-life extends to roughly 6 to 8 days. Without DAC (Mod GRF 1-29), the half-life is roughly 30 minutes and the release pattern is more pulsatile. They are the same core GHRH analog but operate on different timescales.
Can ipamorelin and CJC-1295 be used together?
Yes, and the combination is the most-researched protocol in the GH-axis category. Research has shown the two compounds produce supra-additive GH release because they activate different receptors on the same somatotroph cell. The standard research configuration pairs CJC-1295 (DAC or no-DAC depending on the protocol) with ipamorelin to combine GHRH-receptor amplitude with GHS-R1a frequency.
Why was the CJC-1295 clinical program discontinued?
The phase II program ended after the death of a trial subject. The attending physician concluded the death was most likely attributable to asymptomatic coronary artery disease unrelated to the compound, but the program did not resume. CJC-1295 remains widely studied in research contexts but is not an approved therapeutic.
Which one has more human trial data?
CJC-1295 has the more concentrated human pharmacokinetic dataset, anchored by the Teichman 2006 JCEM trial. Ipamorelin has a broader but more dispersed human dataset across phase I and phase II studies, including bowel motility research. Both are characterised well enough for protocol design; neither is FDA-approved for therapeutic use.
Quantify which one your bloodwork actually points to#
Reading mechanisms is the easy part. Mapping your IGF-1, fasting glucose, sleep architecture, and recovery markers to the right tool is the part that decides outcomes. Run your stats through the Klarovel peptide calculator, complete the research questionnaire, then check how the protocol layer works before you make a single supply decision. The right answer between ipamorelin and CJC-1295 is the one your data points to, not the one the marketing copy points to.
Keep reading
BPC-157 vs TB-500: when to use each for tissue repair
BPC-157 and TB-500 are the two most-researched tissue-repair peptides. Different mechanisms, different use cases. Here's how to decide between them.

Semaglutide vs tirzepatide: the real head-to-head comparison
Semaglutide vs tirzepatide: a direct head-to-head trial settled the efficacy question. Tirzepatide won on weight loss. Here's what else the evidence actually shows.
Melanotan 2 in Norway: Law, Risks, and Safer Tan Options
What research shows about Melanotan 2, why DMP has not approved it in Norway, the documented melanoma case reports, and evidence-based alternatives for pigmentation goals.